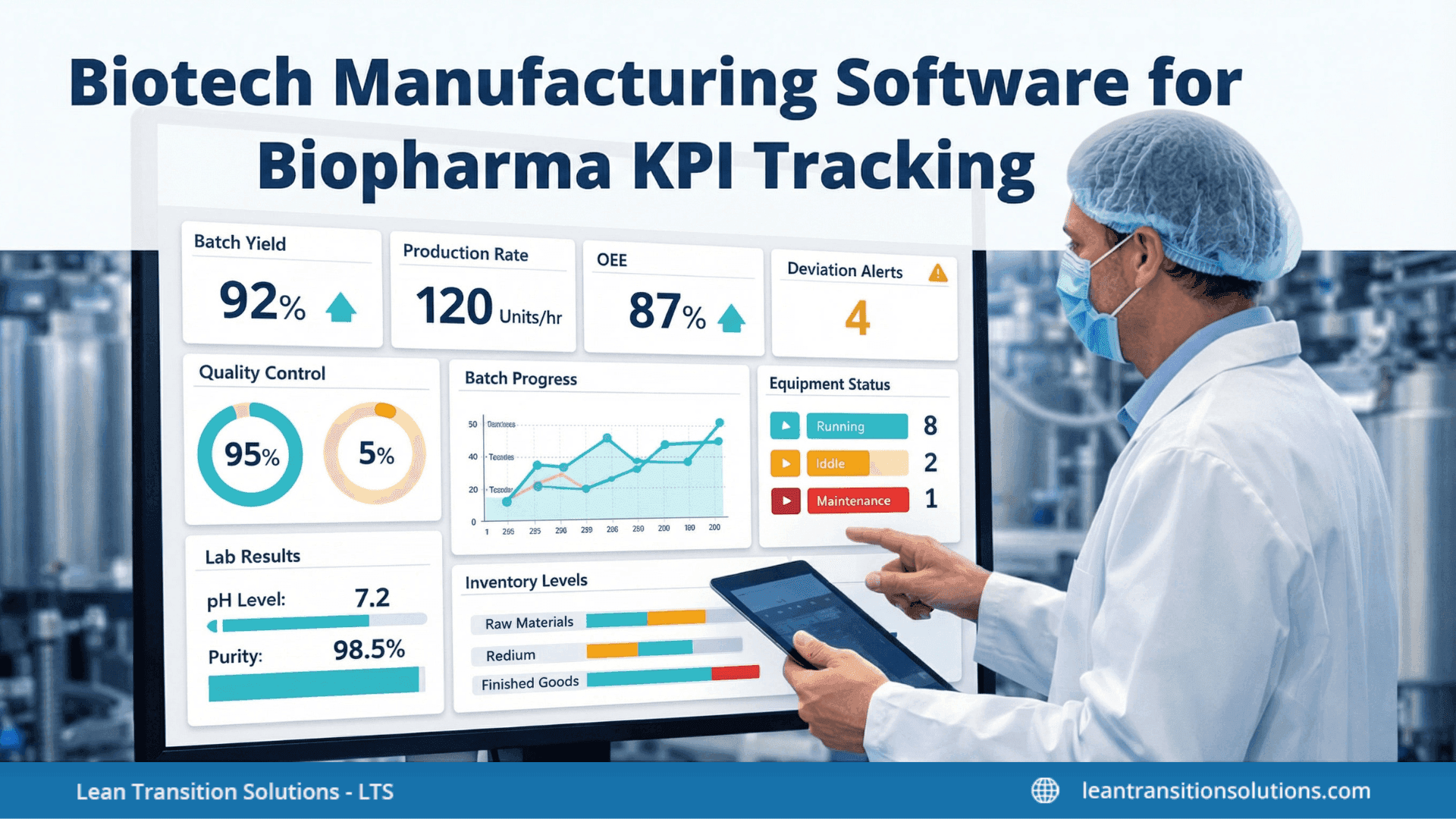
Biotech Manufacturing Software: Powering precision in Biopharma KPIs
Last updated on : March 9, 2026
Let’s be honest — running a biopharma operation today is no walk in the park. Between strict regulations, complex production setups, and the constant push to innovate, it’s easy to feel like you’re always putting out fires. That’s why having the right tools matters. Biotech manufacturing software isn’t just another system — it’s a smarter way to keep track of what’s working (and what’s not). It helps you monitor KPIs in real time, spot issues early, and make decisions based on solid data, not guesswork. Whether you're trying to boost efficiency, stay compliant, or just get a clearer view of your operations, this kind of software can take a lot of the pressure off — and give you back control. In this blog, you will get to know more about what biotech manufacturing software is, why KPI tracking is important in biotech manufacturing, why you will have to integrate biotech manufacturing software with existing pharma systems, and how to choose the right biotech manufacturing software for your industry.
Check out the best customisable biotech manufacturing software solutions by LTS Data Point
What is biotech manufacturing software?
A specialised digital platform called biotech manufacturing software is made to oversee, manage, and improve the intricate production procedures involved in the production of biotechnology and biopharmaceuticals. From upstream and downstream processing to quality assurance and regulatory documentation, it unifies data, workflows, and compliance controls at every level.
Key functionalities
- Live KPI tracking: Monitors biopharma KPIs in real-time enabling leaders to make data-driven decisions.
- Lean Daily Management: Helps team members to track, manage, and enhance their daily task processes using Lean principles using the right LDM tools.
- CAPA: Assists managers to stay one step ahead by spotting issues immediately and providing corrective actions (CA), as well as predict future problems and offer preventive actions (PA).
- Electronic batch records (EBR): Digitally processes data to assure traceability, accuracy, and GMP compliance.
- Manufacturing execution system (MES) integration: Tracks live manufacturing performance and assures action adherence.
- Quality management: Automates deviation, CAPA, and audit management, to sustain product integrity, and meet GMP standards.
- Process analytics: Offers data-driven insights for optimising yields, lowering variability, and enhancing efficiency.
- Equipment and asset management: Monitors calibration, maintenance, and utilisation of critical instruments and bioreactors.
- Inventory and material monitoring: Assures precise control of raw materials, consumables, and finished products.
- Regulatory compliance: Supports FDA, EMA, and other global biotech manufacturing standards such as 21 CFR Part 11 and GAMP 5.
In short, biotech manufacturing software links people, operations, and data across the production cycle, driving operational efficiency, quicker time-to-market, and consistent product quality in highly controlled biotech environments.
Why is KPI monitoring important in biotech manufacturing?
Biotech manufacturing deals with the production of life-saving drugs, biologics, and advanced therapies. These goods need accuracy, consistency, and quality control throughout the manufacturing process. The challenges faced by biotech manufacturing are multi-dimensional, ranging from regulatory compliance to operational efficiency, and each of these challenges can impact both manufacturing costs and patient outcomes. Monitoring biopharma KPIs is necessary for directing these challenges, providing manufacturers the tools to measure, monitor, and optimise their processes in an organised manner.
What are the industry challenges faced by biotech manufacturing?
Getting to know what some of the challenges faced by biotech manufacturing are, can be considered the first and foremost step. Awareness about the current challenges is what drives a leader into deciding if they really need a biotech manufacturing software or not.
Let's see what some of these challenges are.
- Regulatory compliance and quality control: Biotech products are governed by strict regulatory oversight, with agencies such as the FDA, EMA, etc., setting precise standards for manufacturing processes, data integrity, and safety. Any deviation from these standards can cause product recalls, delays, or regulatory fines. Monitoring quality KPIs such as deviation rates or Right-First-Time (RFT) metrics, is necessary for assuring that operations remain compliant and goods meet quality standards.
- Complex manufacturing operations: It includes complex operations such as cell culture, fermentation, etc., which are highly sensitive to variations in conditions such as temperature, pressure, etc. Monitoring KPIs related to these fluctuations enable the employees to get insight on how well the manufacturing process is performing and whether adjustments are needed to minimise fluctuations.
- Supply chain and material management: Biotech products usually depend on high-quality, and at times, rare raw materials. Supervising the supply chain for these materials, especially in a highly unpredictable market, sets forth risks concerning inventory shortages, delays, and quality issues. Biopharma KPIs such as inventory turnover, lead time, and material usage efficiency assists in tracking the flow of materials, assuring that manufacturing schedules are met, and the right materials are available when required, all while avoiding excess inventory or stockouts.
- Cost control and efficiency: Biotech manufacturing is an expensive process as it deals with specialised equipment, skilled labour, and raw materials. Manufacturers are under constant pressure to lower costs without compromising on quality and consistency due to the intensifying competition. Biopharma KPIs such as cost per unit produced, cycle time, and capacity utilisation aids in tracking manufacturing costs, detect inefficiencies, and offer data-driven opportunities to optimise resource allocation and minimise waste.
- Scalability and flexibility: The rising demand for biotech products – in areas like gene therapies, vaccines, biologics – poses scalability challenges. Manufacturers need to maximise production without compromising on quality or efficiency. Biopharma KPIs that monitor on-time delivery and batch release times are necessary for managing production scale-ups, assuring that the system can handle increased volume without sacrificing quality or time.
- Risk management and traceability: A small fault in manufacturing process can lead to financial and reputational damage. This traceability is also crucial for detecting and reducing risks. Biopharma KPIs like deviation reporting, audit readiness, and non-conformance rates allow manufacturers to stay on top of potential risks, ensuring that they can take immediate corrective actions before a problem escalates.
Why KPI tracking is important in biotech manufacturing
In biotech manufacturing KPIs are not some random numbers, instead they are productive insights that help manufacturers make data-driven decisions, sustain control over their operations, and direct the complexities of the organisation. This is where proper tracking of biopharma KPIs becomes necessary.
- Assures regulatory compliance: Biotech manufacturing must fulfil exact standards set by regulatory bodies. By monitoring key metrics like deviation rate, product quality, and audit outcomes, KPIs help confirm compliance with regulations like good manufacturing practices (GMP). Tracking these KPIs actively enables manufacturers to take corrective action before an issue escalates resulting in fines or product recalls.
- Enhances operational efficiency: KPIs such as cycle time, capacity utilisation, and overall equipment effectiveness (OEE) offer real-time data on manufacturing processes, assisting in spotting bottlenecks, downtime, and inefficiencies. These insights enable manufacturers to organise functions, minimise waste, and maximise the use of costly resources and equipment.
- Optimises resource allocation: KPIs related to labour efficiency, material utilisation, and equipment uptime offer manufacturers with a clear idea on how resources are being used. These metrics enable manufacturers allocate resources more conveniently, enhancing profitability without sacrificing product quality.
- Motivates continuous improvement: Calculating KPIs often enables manufacturers to spot areas that need improvement, implement changes, and then compute the impact of those changes over time. KPIs like process capability, and yield improvement assist in tracking the success of functional changes and encourage a culture of operational excellence.
- Allows real-time tracking and quick decision-making: With live KPI monitoring, manufacturers can make data-driven decisions quickly, addressing problems as they escalate. This responsiveness is critical to maintaining manufacturing schedules, assuring product quality, and preventing costly delays or bottlenecks.
- Improves product quality and safety: Quality KPIs such as RFT rate, deviation rate, and lot acceptance rate makes sure that the manufacturing processes remain under control and that each batch produced fulfils quality standards. Monitoring these KPIs allows manufacturers to identify problems early and make necessary adjustments before they impact the final product or lead to recalls.
- Maintains scalability and flexibility: Tracking KPIs like inventory turnover, and supply chain efficiency offer how well the business is managing progress, aiding in preventing the growing pains that can happen when expanding operations.
- Helps in minimising risk: Monitoring KPIs related to deviation rates, non-conformance, and audit outcomes enables manufacturers to detect potential risks in the manufacturing process and establish corrective actions before issues escalate. This proactive approach lowers the chance of producing non-compliant batches, avoiding expensive rework or product recalls.
Key features and benefits: What makes biotech manufacturing software perfect for KPI management
Biotech manufacturing software is specifically designed to improve KPI visibility, organise functions, and assist data-driven decision-making. These systems offer a combined platform for tracking performance across all phases of manufacturing by integrating advanced features like live analytics, dashboarding and compliance monitoring. This results in significant efficiency, enhanced accuracy, and stronger regulatory alignment.
Key features of biotech manufacturing software that improve KPI visibility
- Interactive dashboards: Visual dashboards behave as intuitive by bringing every key performance indicator into a single screen, enabling stakeholders to track manufacturing, quality, and efficiency metrics in real time. This assists in spotting trends, oddities, and performance gaps immediately.
- Live analytics: Leverages real-time reports from equipment, sensors, and manufacturing systems to offer fast decision-making and faster issue resolution.
- Compliance and audit tracking: Makes sure that all KPI data is aligned with organisation regulations such as FDA 21 CFR Part 11, GMP, and GAMP 5. It sustains a detectable record of activities, deviations, and corrective actions, simplifying compliance audits and minimising risk of regulatory breaches.
- Automated data collection and integration: Flawless integration with MES, LIMS, ERP, and other systems automates data capture across manufacturing lifecycle. This puts a stop to manual entry errors and assures that KPI monitoring reflects precise, up-to-date information from every stage of production.
- Customisable data and alerts: Users can frame alerts for KPI outliers, set performance thresholds, and create tailored reports for different roles or departments. This customisation assures that each team has the most important details to make timely decisions.
- Historical data and trend analysis: The capacity to evaluate historical performance trends aids manufacturers detect repeating problems, predict future results, and encourage continuous improvement strategies.
Benefits of biotech manufacturing software
- Enhanced operational efficiency: Automated monitoring and live visibility lowers manual tracking efforts and manufacturing downtime helping teams quickly spotting inefficiencies, optimise resource allocation, and improve overall process performance.
- Improved accuracy and data integrity: Reduces manual error and makes sure that KPI reports are reliable, consistent, and audit ready by automating data collection and authentication.
- Regulatory alignment and audit readiness: Built-in compliance management verifies that all KPI report aligns with regulatory requirements. The system’s ability to track and document makes it easier to exhibit compliance during inspections or audits.
- Faster and better decision-making: With live analytics and immediate access to performance data, managers can make informed decisions quickly. This allows industries to tackle deviations instantly, capitalise on performance insights, and drive continuous improvement.
- Scalability and standardisation: Secures that KPI tracking remains consistent and scalable across multiple facilities, product lines, or geographies even as the company shows constant growth.
In short, biotech manufacturing software modifies KPI management from a passive process to a proactive and data-driven strategy. It empowers industries to attain greater efficiency, precision, and regulatory confidence resulting in better performance and sustainable growth.
Why you need to integrate biotech software with existing pharma systems
Integrating biotech manufacturing software with existing pharma systems – MES, ERP, and LIMS – is necessary for attaining flawless data flow, operational efficiency, and unified performance tracking. As biotech manufacturing involve complex workflows, interconnected systems make sure that every stage is aligned, trackable, and optimised for regulatory compliance.
1. Integration with manufacturing execution system (MES): Acts as the bridge between enterprise-level planning and shopfloor execution. Integrating biotech software with MES enables real-time visibility into production activities, equipment status, and process frameworks.
Benefits:
- Allows automated gathering of reports from the production floor.
- Makes sure that there is synchronisation between batch records, operation control, and KPI monitoring.
- Minimises manual recording and secures data integrity in alignment with GMP standards.
2. Integration with enterprise resource planning (ERP): ERP systems are in control of business-wide functions such as ownership, inventory, and financials. Integrating with ERP offers an all-inclusive view of both manufacturing and business performance.
- Coordinates material requirements, production schedules, and financial planning.
- Enhances detectability from raw material acquisition to finished products.
- Streamlines resource allocation and expenditure monitoring for better operational control.
3. Integration with laboratory information management system (LIMS): LIMS supervises laboratory workflows, sample monitoring, and test results which are some of the vital components of biotech quality management. Integration with biotech software verifies that laboratory and manufacturing details are linked for end-to-end quality oversight.
Benefits:
- Automatically transfers analytical data and test results to production systems.
- Minimises unnecessary data entry and the risk of recording errors.
- Improves batch release efficiency by connecting lab results with manufacturing KPIs.
4. Ensuring data interoperability: For successful integration, data compatibility is critical – enabling different systems to communicate using standardised report formats and protocols.
Best practices:
- Use open APIs or middleware platforms to ease seamless data exchange.
- Adopt industry standards like ISA-95 and OPC-UA for system linking.
- Secure data harmonisation so that KPI definitions and calculations remain consistent across systems.
- Execute strict data governance to maintain accuracy, security, and compliance.
How to choose the right biotech manufacturing software for your biopharma industry with LTS Data Point?
If you are looking for a biotech manufacturing software that is not rigid but is flexible enough to fulfil your needs, choosing LTS Data Point software solutions would be your best strategic move. Let's explore what makes LTS Data Point unique in this field.
Key features
- Configurable dashboards: Real-time KPI monitoring across core dimensions such as Safety, Quality, Delivery, Cost and People (SQDCP), with customisable dashboards at shift, plant or enterprise level.
- Strategic alignment: Built-in support for frameworks like FCIL (Financial, Customer, Internal, Learning and growth) and Balanced Scorecard, allowing mapping of strategy, metrics, and action.
- Interactive dashboards and strategy maps: Visual tools for connecting high-level goals with operational KPI performance and enabling drill-down.
- Visual management and continuous improvement support: Modules for root-cause analysis, KPI improvement workflows, value-stream mapping, digital huddle boards.
- Multi-site scalability: Ability to standardise KPIs across sites, compare performance globally yet adapt locally.
- Integration readiness: With smooth connectivity to ERP and MES systems, biotech manufacturing software secures centralised, real-time access to critical KPIs.
How can LTS Data Point become beneficial for your biotech manufacturing business?
- Operational transparency and informed decisions: With real-time dashboards and KPI visibility, teams can quickly detect issues – deviation trends, low yield – and tackle proactively.
- Strategic-to-execution alignment: The tool helps assure that shopfloor metrics link to corporate goals.
- Continuous improvement enablement: Fixed Lean/CI modules mean you don’t just monitor KPIs, you also manage improvement actions — accelerating yield gains, waste minimising, process stability.
- Scalability across manufacturing sites: For biotech businesses with multiple plants or global footprints, a systematised KPI platform aids maintain consistency in performance and reporting.
- Reduced data silos and improved data integrity: By integrating with existing systems (MES/ERP/LIMS), the KPI tool makes sure calculation is based on trusted, timely data — enhancing accuracy and audit readiness.
LTS Data Point brings to life, your dream about running your biotech manufacturing company without any friction. Let's also look into real-world experiences of biopharma companies who utilised software solutions offered by LTS Data Point.
Real-world examples of biopharma companies using LTS Data Point biotech manufacturing software: Is it worth your money?
Real-world examples of pharma companies utilising customisable biotech management software helps you get an idea on how this works. Whether your company is primarily pharmaceuticals or biopharmaceuticals, the methods are almost the same. If it’s not and you need customisation, that’s where you need to find a flexible software solution like LTS Data Point that supports your company’s needs.
Real-world example one: How a pharma company that faced challenges of manual reporting and siloed KPIs tackled it with LTS Data Point pharmaceutical manufacturing software
At a pharmaceutical manufacturing site, the organisation replaced different manual reporting and siloed KPIs with a unified digital Balanced Scorecard (BSC) system through Data Point. The solution combined real-time data with production, quality, supply chain and cost domains, allowing leadership to visualise strategy-to-execution alignment, quickly spot performance gaps and streamline decision-making. By shifting to this integrated view, the company improved operational transparency, enhanced performance tracking and offered managers with productive insights rather than raw data, thereby supporting sustained performance improvement and regulatory readiness.
See our blog for more details: Pharma Manufacturing | KPI & Compliance with Data Point
Real-world example two: How PCI Pharma’s Bridgend facility tackled their challenges with LTS Data Point pharmaceutical manufacturing software
At PCI Pharma’s Bridgend facility, traditional paper-based and spreadsheet methods for daily stand-up meetings and tracking metrics across Safety, Quality, Delivery, Cost and People (SQDCP) were replaced by a fully digital balanced scorecard using Data Point. This shift enabled real-time visibility of KPIs, streamlined communication of strategic goals, and aligned day-to-day operations more tightly with the site’s vision. As a result, the site achieved clearer process-alignment, improved data capture and analysis, better-informed action-planning, and enhanced continuous improvement capability across all key operational domains.
For more details, read our blog: Streamlining Continuous Improvement | Data Point | PCI Pharma
To sum up, if you're working in biotech manufacturing company and still relying on spreadsheets or disconnected systems to track KPIs, it might be time for a smarter approach. Biotech manufacturing software doesn’t just simplify things; it gives you real-time visibility, sharper insights, and the confidence to make quick, informed decisions. With smooth integration into your existing ERP, MES, and LIMS systems, it brings all your performance data together in one place. Whether you're aiming to boost compliance, speed up production, or just get clearer visibility across operations, this software could be the game-changer your team’s been waiting for.
Join hands with LTS Data Point for a smarter future
FAQs
1. What is biotech manufacturing software used for in pharma?
Biotech manufacturing software helps pharmaceutical companies manage production processes, track key performance indicators (KPIs), and ensure compliance with industry regulations.
2. Why is KPI tracking important in biopharma manufacturing?
KPI tracking enables biopharma companies to monitor efficiency, quality, and compliance, helping improve decision-making and reduce operational risks.
3. Can biotech software integrate with ERP and MES systems?
Yes, leading biotech software solutions offer seamless integration with ERP, MES, and LIMS systems to centralise performance data and streamline workflows.
4. How does biotech software support GMP and regulatory compliance?
It automates documentation, tracks deviations, and maintains audit trails, helping meet standards like GMP, FDA 21 CFR Part 11, and GAMP 5.
5. What types of KPIs can be monitored using biotech manufacturing software?
You can monitor KPIs such as production yield, deviation rates, inventory turnover, cycle time, and equipment effectiveness — all in real time.
6. Is biotech manufacturing software suitable for multi-site pharma operations?
Absolutely. It supports multi-site scalability, allowing consistent KPI tracking and performance benchmarking across different locations.
7. How does real-time KPI tracking improve biopharma operations?
Real-time tracking helps teams identify issues quickly, make informed decisions, and take corrective actions before problems escalate.
8. What are the benefits of switching from manual KPI tracking to software?
You’ll gain improved accuracy, faster insights, better compliance, and reduced downtime — all while saving time and resources.
9. Can the software be customised for specific biopharma workflows?
Yes, most biotech software platforms offer configurable dashboards, alerts, and reports tailored to your organisation’s unique needs.