RCCM in Manufacturing: A Smarter Way to Identify Root Causes and Drive Corrective Action

Last updated on : April 29, 2026
The same problems keep popping back up. That is not bad luck – it is a system that never actually fixed anything.
Most manufacturing teams respond fast when something goes wrong. The line stops, the defective batch gets pulled, the operator gets a reminder. And three weeks later, the same problem is back.
RCCM – root cause and corrective measures – is the structured discipline of tracing failures to their true systemic origin and implementing measures that permanently eliminate the cause, not just manage the symptom. It exists to break exactly that cycle. Without it, teams remain busy reacting to problems they have already “solved” once before.
Root cause analysis in manufacturing is not about working harder on quality. It is about making sure the work you do actually sticks.
See how LTS Data Point helps manufacturing teams track root causes and close corrective actions faster
What RCCM actually means – and what it does not
Root cause and corrective measures (RCCM) is not troubleshooting. That distinction matters more than most teams realise. The simplest way to tell them apart is by the question each one asks.
Troubleshooting: How do we get running again?
RCCM: Why did this happen, and what needs to permanently change?
A correction replaces or reworks the defective output. A corrective measure eliminates the process condition that produced it.
These two might sound similar in the beginning, but over time, the difference between them shows up in whether your problem list shrinks or simply rotates.
Three things RCCM is not:
- A compliance form to complete after an incident
- A formal way of pointing at the nearest operator
- A one-off quality audit activity
What RCCM actually is:
- A structured, repeatable method for diagnosing process failures
- A system for assigning clear ownership to permanent fixes
- A discipline that builds genuine problem-solving capability across the organisation
The organisations that treat RCCM as just a reporting tool tend to see the same problems recycling quarter after quarter. Those that treat it as an operating discipline see their problem list genuinely shrink.
How the RCCM process works: Step by step
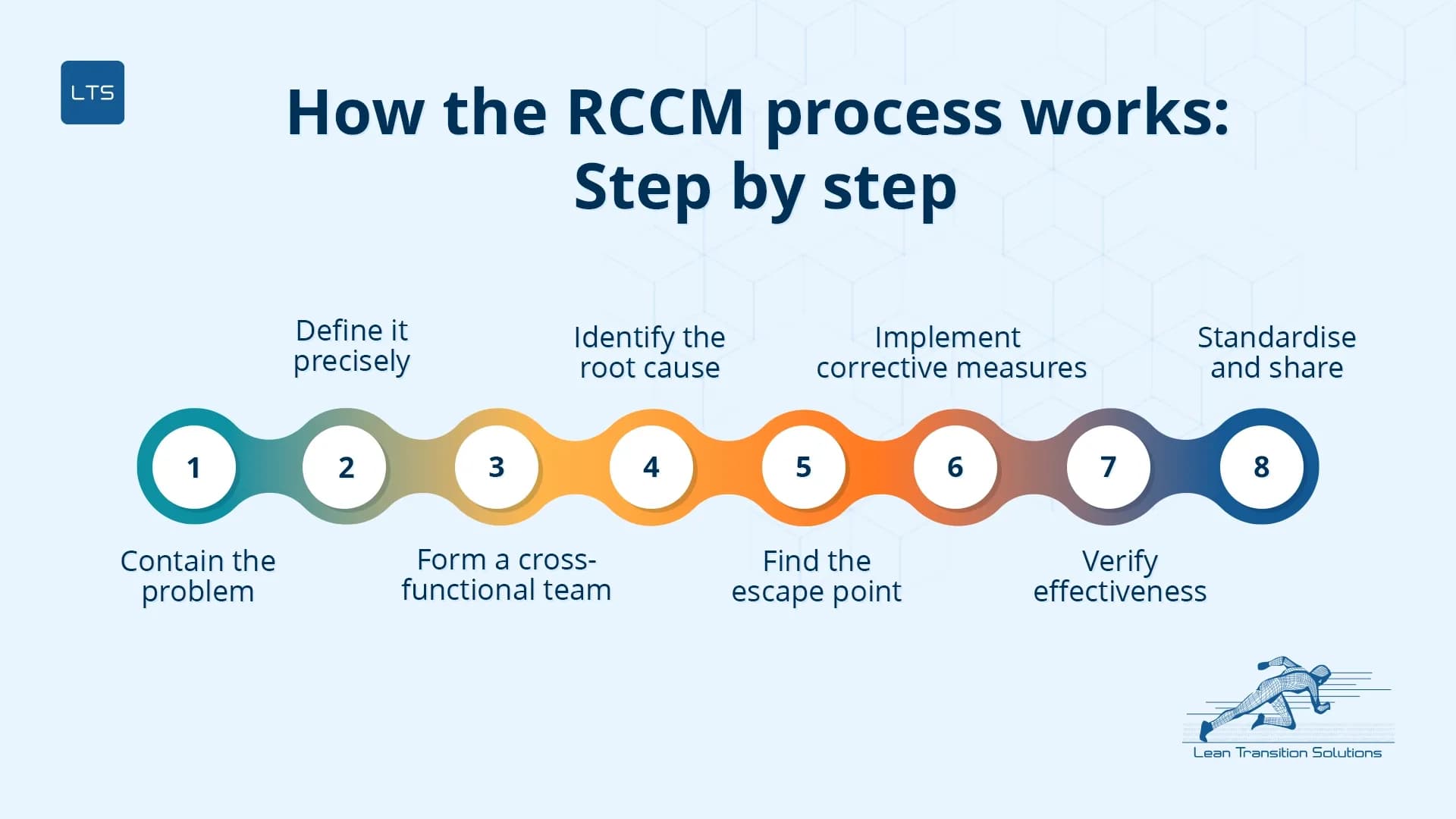
The RCCM process follows a clear, structured sequence – one that aligns with the 8D problem-solving methodology. This methodology is widely adopted across automotive and regulated manufacturing and reflects the logic of PDCA cycle thinking.
Contain the problem
Stop the immediate bleed. Quarantine affected product, isolate the process, protect the customer from further impact.
Define it precisely
Use data, not assumptions. Specify what failed, where, when, and how frequently.
Form a cross-functional team
Operators, engineers, and quality leads together. The people closest to the process hold the most useful information.
Identify the root cause
Use 5 Whys analysis or a Fishbone diagram approach. Keep asking why until you reach a cause within management’s control to permanently fix.
Find the escape point
Identify where the process should have caught the problem but did not. Address both the cause and the detection gap.
Implement corrective measures
Target the root cause directly. Update SOPs, control plans, and work instructions accordingly.
Verify effectiveness
Confirm the problem has actually stopped. Define a monitoring window and success criteria before closing the record.
Standardise and share
Deploy lessons learned to similar lines, cells, or sites so they fix compounds across the operation.
Each step builds on the one before it – containment buys time, investigation finds the truth, and implementation delivers the fix. But the cycle is only complete when step seven is done properly.
Closing a record and verifying an outcome are two entirely different things. Most teams treat them as the same thing. When that happens, the corrective action exists on paper, but the problem continues on the floor.
Why corrective actions fail to stick
There is a pattern most manufacturing teams will recognise immediately – teams work hard on quality, but recurring failures never seem to reduce.
The problem is rarely effort. It usually is one of the following three things:
- Investigations stop too early: When root cause findings consistently point to human error, that is almost always a sign the analysis did not go deep enough. Human error is a symptom, not the cause. The process condition that made the error easy to make is the root cause countermeasure that was never found.
- No one owns the outcome: A corrective action without a named owner and a defined due date is a suggestion. Teams under production pressure will prioritise output over follow-through – not because they do not care, but because nothing is holding the action open.
- Effectiveness is never confirmed: Most corrective action management processes close records when the action is implemented – not when the problem is confirmed resolved. Those are different moments and treating them as the same is how failures quietly return.
Most teams know this is happening. Very few have a system that stops it.
The real damage happens when your best people spend every day putting out fires that should never have started.
Investigations end up getting rushed because there is always something more urgent. Follow-through disappears because production pressure wins every time. And silently, without anyone deciding it, the plant starts celebrating the person who saved the shift – not the one who stopped the problem from happening in the first place.
That is not a people problem. That is a culture that manufacturing quality improvement cannot survive in.
RCCM inside lean daily management: Where it actually lives
RCCM does not live in a quality department database. It lives in the daily rhythm of the plant.
In a plant that runs lean daily management properly, RCCM shows up at every tier – not as a report, but as a conversation.
At Tier 1 shift huddles, teams review the SQDCM board. Any metric in red triggers a structured conversation – what happened, what containment is in place, and whether a formal investigation is needed. Actions leave the meeting with named owners and due dates.
At Tier 2, escalated issues receive cross-functional attention.
At Tier 3, persistent or systemic problems are addressed at plant leadership level with the resources they need.
Gemba walks reinforce the cycle. Leaders observe work at the point it happens, review open corrective actions on visual boards, and confirm that implemented fixes are holding. Shopfloor problem solving is embedded in every tier of the daily review.
The board’s job is not to show green. Its job is to surface the conversations that green can hide.
How LTS Data Point supports structured RCCM
Think about a quality lead starting their shift, opening their dashboard, and spotting a deviation trend before the morning huddle even begins – that is what structured digital RCCM looks like in practice.
But structure alone is only as strong as the system that supports it. When corrective action management runs on spreadsheets, verification steps get skipped, actions go overdue without escalation, and lessons learned stay trapped on a single site.
LTS Data Point gives manufacturing teams one place where every stage of the RCCM cycle – from first detection to verified closure – is visible and accountable.
- KPI deviation detection: Live dashboards and visual scorecards flag performance gaps as they emerge. Quad chart analysis combines trend data and narrative context in one view, so leaders arrive at conversations with facts rather than gut feel.
- Root cause analysis: Dynamic Fishbone diagrams support structured non-conformance management directly within the platform. Pareto charts surface recurring failure patterns so corrective effort is focused where it has most impact.
- Corrective action tracking: A centralised log assigns owners, sets deadlines, and tracks progress in real time. Automated reminders and escalations ensure actions do not quietly expire between meetings.
- Effectiveness verification: Closure requires documented evidence that the corrective measure achieved its defined outcome. The platform does not permit premature closure – the step most manual processes skip entirely.
- Lessons learned: Resolved cases feed into a documented knowledge base, enabling deployment across lines and sites rather than keeping fixes local.
Teams managing CAPA in manufacturing through LTS Data Point move from reactive incident response to a structured improvement cycle that compounds over time. As the time passes the organisations stops reacting to the same problems and starts solving new ones. That is what improvement actually looks like.
Most plants already have the methodology. What they are missing is the follow-through.
The root cause gets found. The action gets assigned. And then production pressure takes over, the record gets closed, and three months later the same problem is back on the board. That gap – between doing the work and verifying it actually worked – is where most improvement effort quietly disappears.
RCCM closes that gap. Not by adding more process, but by making accountability visible at every tier – from the shift huddle to the plant director’s review. When teams feel safe raising problems early, when every corrective action has a named owner and a verified outcome, and when lessons learned travel across sites instead of staying local – that is when a manufacturing organisation stops managing its failures and starts genuinely reducing them.
LTS Data Point gives every layer of the organisation the structure to make that happen – not occasionally, but every single day.
Your next recurring failure is already in motion – talk to an LTS Data Point expert today before it costs you another cycle
FAQs
1. Is RCCM suitable for small and mid-sized manufacturers only large plants?
RCCM is not scale-dependent. The structured thinking – define the problem, find the true cause, verify the fix – applies equally to a 50-person facility and a multi-site operation. The tools and formality may differ, but the discipline is the same.
2. How long should a monitoring window be after implementing a corrective measure?
There is no universal answer – it depends on how frequently the problem occurred. A general rule is to monitor for at least as long as it would take the problem to reasonably recur under normal operating conditions. For high-frequency issues, two to four weeks may be sufficient. For lower-frequency failures, three months or more may be needed.
3. Can RCCM be applied to non-production problems such as delivery delays or supplier failures?
Yes. RCCM applies to any repeating performance gap – delivery, procurement, planning, or supplier quality. The process is the same: define the deviation, trace the systemic cause, implement a permanent measure, and verify the outcome.
4. What is the difference between RCCM and A3 problem solving?
Both are structured problem-solving approaches, but A3 is broader – it encompasses problem definition, analysis, countermeasures, and a communication format on a single page. RCCM focuses specifically on root cause identification and the corrective action cycle. In practice, an A3 often serves as the documentation format for an RCCM investigation.
5. How do you prioritise which problems to run an RCCM on?
Not every issue warrants a full RCCM investigation. Focus on problems that are recurring, that have a measurable impact on safety, quality, delivery, or cost, or that have previously been “closed” without resolution. A Pareto analysis of your top defect or failure categories is a practical starting point.
6. Who should own the RCCM process – quality, operations, or engineering?
Ownership of the overall RCCM system typically sits with quality or continuous improvement, but individual corrective actions should be owned by whoever controls the process where the root cause lives – which is often operations or engineering. Assigning every action to the quality team is one of the most common reasons corrective actions fall to drive real change.