Pharmaceutical Manufacturing Software That Turns Lean Daily Management into Operational Excellence
Most pharma sites track KPIs. Data Point closes the loop. It is the pharma lean management platform that turns that data into structured daily action - connecting your operations, quality, and strategy teams under one pharmaceutical continuous improvement system built for regulated environments.
Pharmaceutical Manufacturing Software Built for Daily Management, Quality, and Strategy
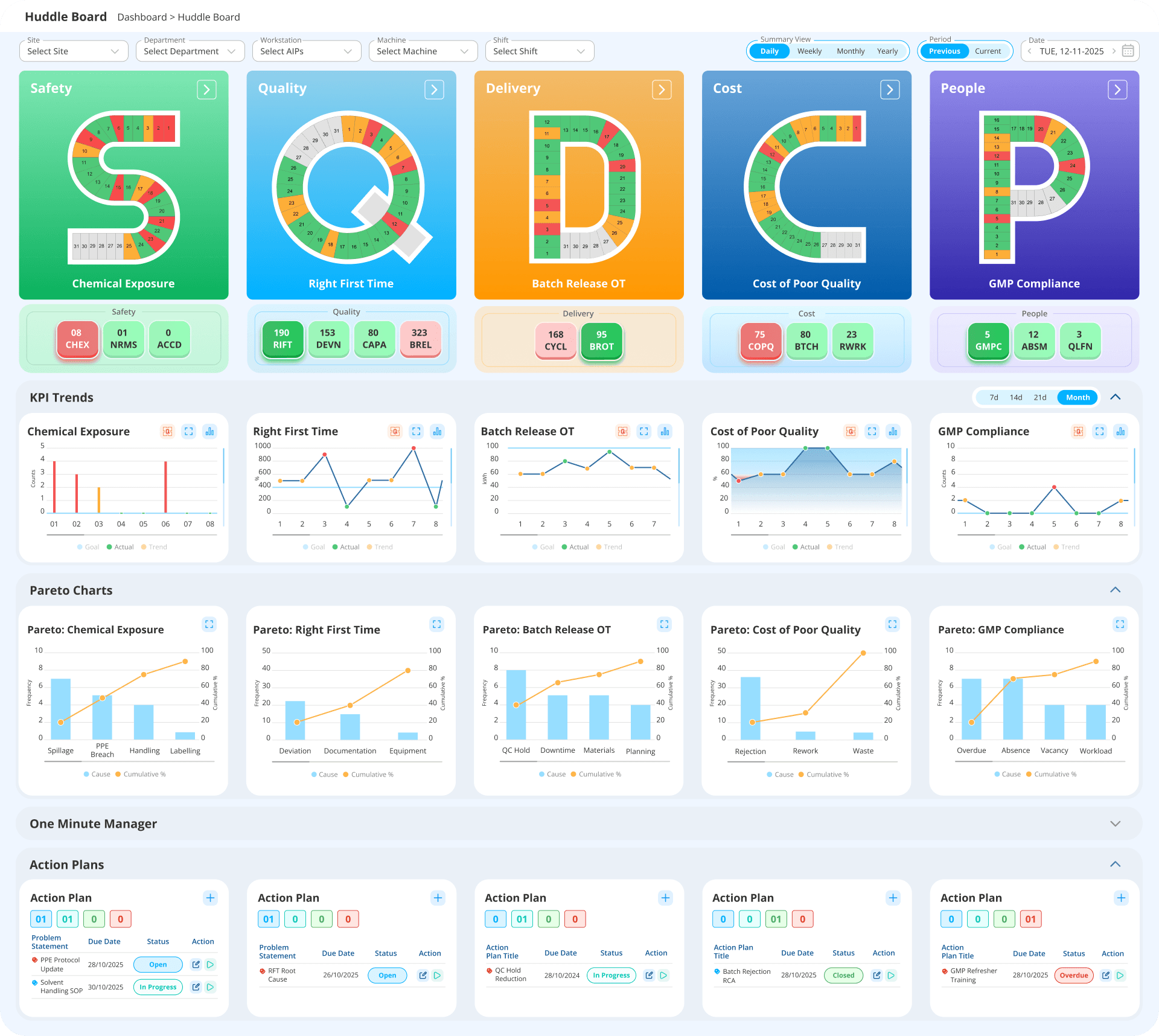
Real-time SQDCP performance, open CAPA actions, and improvement status - visible to every tier, updated live from your connected systems.
Choice of industry leaders and Fortune 500 companies








































































Everything Pharmaceutical Operations Needs. One Connected System.
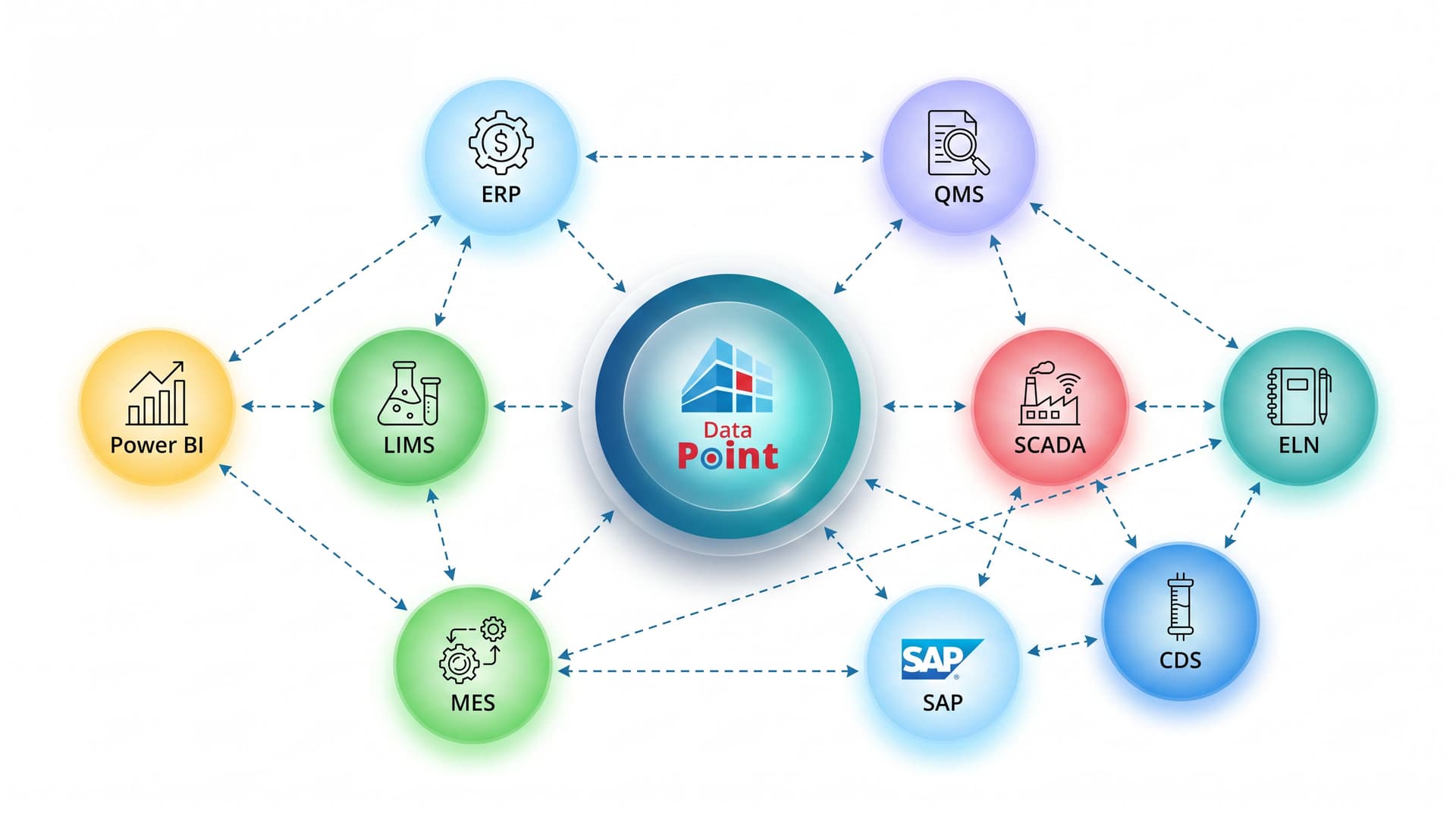
LTS Data Point is a Pharma manufacturing software platform where every module connects to the next. Your SQDCP tier board surfaces a quality gap, that gap triggers a CAPA. The CAPA links to a root cause investigation and the fix feeds into a digital VSM improvement. The whole programme rolls up into your Hoshin Kanri strategy. No data silos, no disconnected tools.
SQDCP Daily Management
A Pharma daily management system (SQCDP) for pharmaceutical manufacturing. It replaces paper DDS (Daily Direction Setting) boards with live tier boards - giving every level from shift leader to site director real-time SQDCP visibility with RAG status driven by live data. Supports SQDCP, SQDCM, SQODCPI, and fully custom frameworks.

CAPA & Deviation Management
As CAPA management software pharmaceutical operations teams rely on, Data Point embeds this pharma CAPA tracking system directly into your daily performance cycle; not isolated in a standalone QMS. When a quality KPI turns red, a CAPA is raised, tracked through the 4C's workflow, and auto escalated if it slips.
Hoshin Kanri Strategy Deployment
Hoshin Kanri pharma manufacturing deployment means your annual breakthrough objectives stay connected to what shift teams are actually doing - every week of the year. Data Point's digital X-Matrix cascades strategy from site leadership to team-level PDCA actions, tracked live with Quad Chart and One Minute Manager tools.

Digital Value Stream Mapping
Go beyond drawing maps. Data Point's Digital VSM classifies each process step as VA, NVA, or BVA automatically, calculates Process Cycle Efficiency, and flags your highest-NVA bottleneck in real time. Run "what-if" simulations before committing resources, then link every improvement directly to your X-Matrix strategy.
Root Cause Analysis & Problem Solving
A complete pharmaceutical continuous improvement software toolkit - embedded in your quality and operations workflow. Every investigation is structured, traceable, and linked to the CAPA it addresses. Choose from 5 Why, Fishbone, 8D, PDCA, Mind Map, or Process Flow within the same workflow.

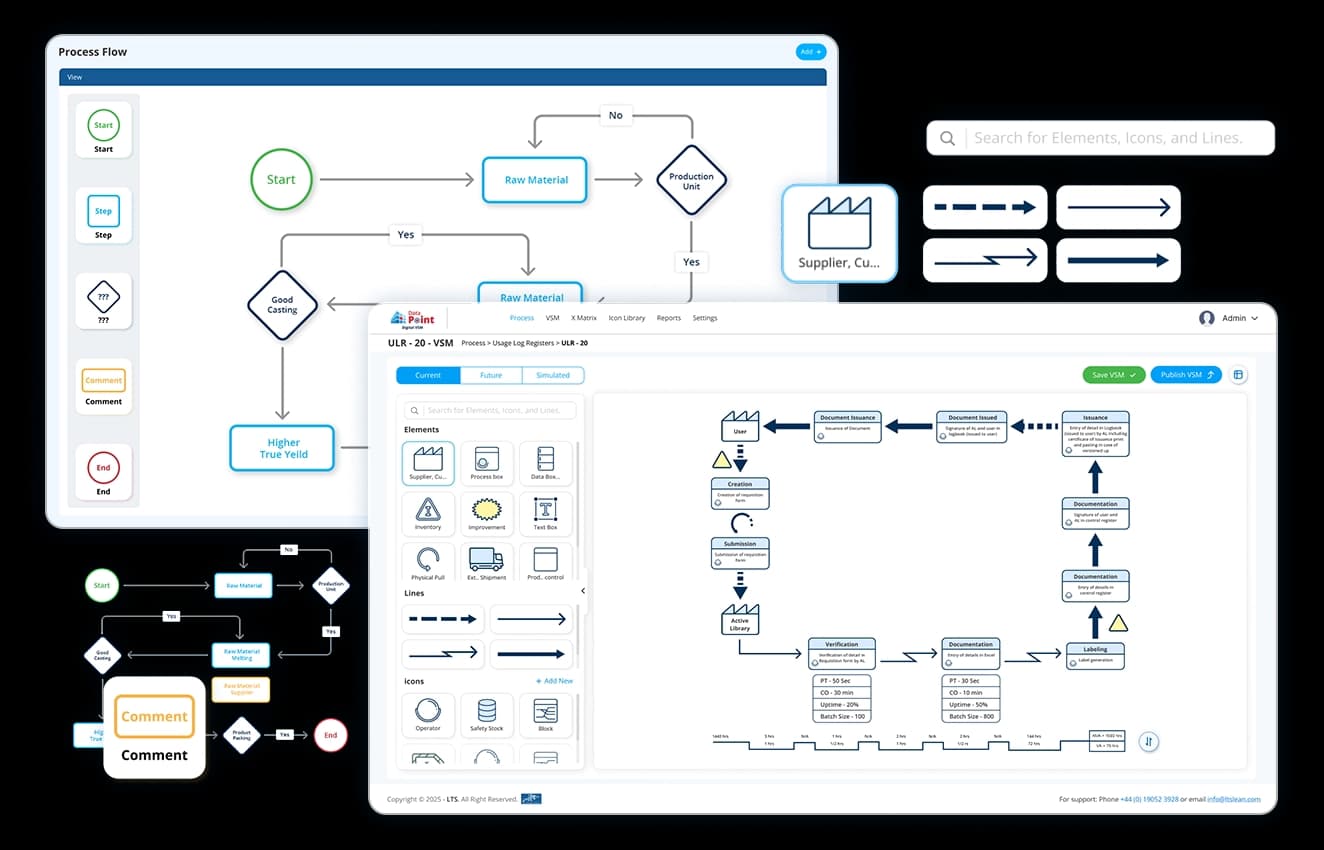
System Integration & Live Data
Data Point connects to your existing ERP, LIMS, and MES systems - pulling production, QC, batch, and cost data directly into your SQDCP tier boards. No weekly exports, no copy-paste. Your morning huddle runs on today's numbers. Manual input is fully supported where direct integration is not yet in place.
Operational Accountability Across Every Team, Site, and Action
Three additional capabilities that complete the Data Point picture - connecting people, actions, and sites into one accountable performance system.
Track analyst qualifications, competency levels, and training completion across production and QC teams. Identify skill gaps before they cause batch delays or capacity issues. QC labs can connect with LIMS to see analyst productivity, sample turnaround time, and shift workload in real time; so, resource planning is based on live data, not a spreadsheet updated once a week.
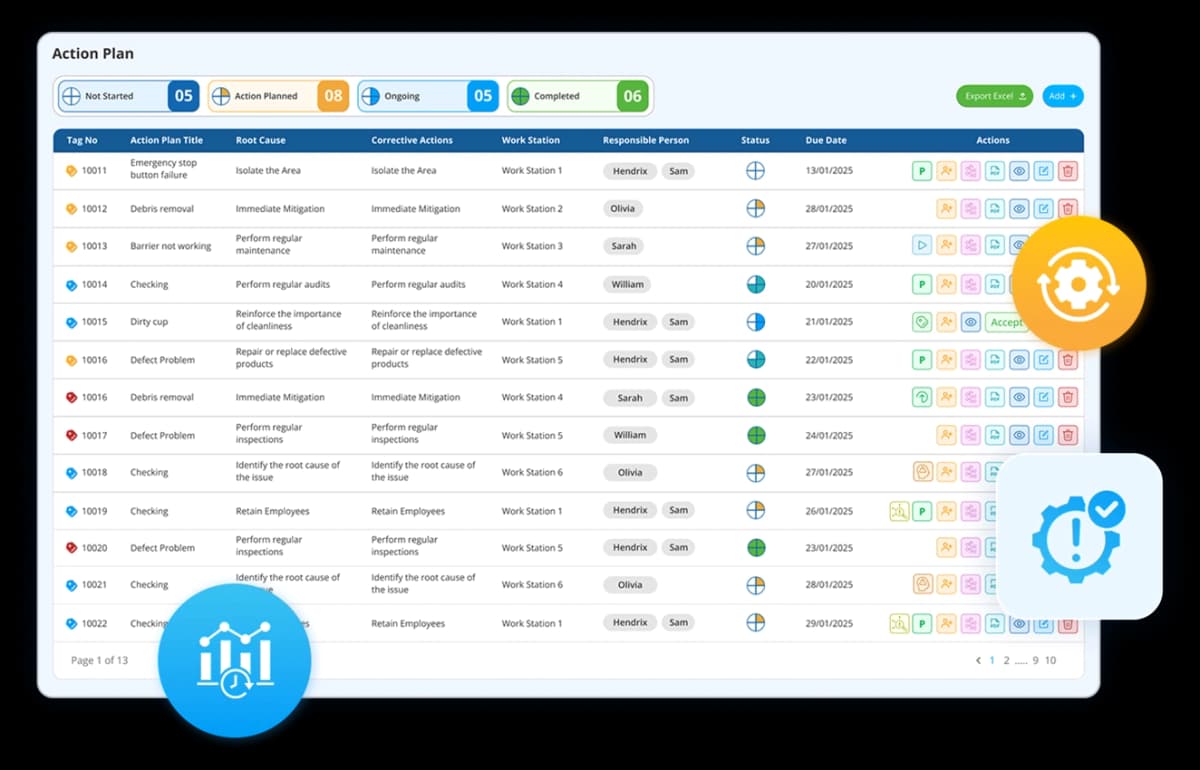
Every open action across every department, visible in one place. Owner, due date, and status tracked automatically. Actions from tier board huddles, investigations, and strategy reviews all land in the same plan so nothing is missed. Overdue actions escalate without anyone having to ask.
Use the same daily management framework and KPIs across every site, with one group-level view for leadership. Start with a single line or site, prove the approach works, then roll it out further at your own pace. No new infrastructure needed. Cloud-hosted and configured to your organisation's structure from day one.
From Visual Management to Pharmaceutical Process Improvement.
Before LTS Data Point, getting a clear view across our sites meant chasing reports and reconciling numbers. Now, with Data Point as our pharmaceutical manufacturing software, everything is visible in real time. We can see batch performance, site KPIs, and issues as they happen, all in one place. What's made the biggest difference is how quickly we can act. Leadership reviews are no longer about explaining last week's data. We focus on decisions, priorities, and next steps. It has brought a level of clarity and alignment we simply didn't have before.
COO
Pharmaceutical Contract Manufacturing Services, UK
Deviation tracking used to be reactive and fragmented. We were constantly following up across systems just to understand status. With Data Point, every deviation, investigation, and CAPA is visible in real time, all in one place. What stands out is the level of control we now have. Nothing gets missed, ownership is clear, and progress is always visible. It has significantly improved our audit readiness and given us much stronger confidence in our quality processes.
QA Director
Biopharmaceutical Manufacturing, EU
Daily management has completely changed. Instead of waiting for reports, we see line performance and issues as they happen. Teams take ownership faster, and we act on problems immediately.
Plant Manager
Oral Solid Dosage (OSD) Manufacturing Facility, India
See LTS Data Point Pharmaceutical daily management system in a live pharma environment
Book a DemoWhat Pharmaceutical Organisations Achieve with Pharma Manufacturing Software
Pharmaceutical Manufacturing KPI Tracking delivers real operational improvements for Pharma operations and biopharmaceutical sites by replacing manual systems and disconnected tools.
Live dashboards replace manual slide compilation. Reviews run on today's data, not last week's.
Operations teams see KPI performance in the same shift it happens, not the following morning.
Digital VSM pinpoints your biggest NVA bottlenecks and simulates improvements before you act.
LTS Data Point vs Traditional Pharma Operations Management
Most pharmaceutical sites manage operations through a combination of paper boards, disconnected spreadsheets, and standalone systems that don't talk to each other. Data Point is the pharmaceutical lean manufacturing software platform that replaces that fragmentation with a single connected system; where every improvement is visible, owned, and traceable.
| Capability | Traditional Pharma manufacturing management system | LTS Data Point Pharmaceutical manufacturing software | |
|---|---|---|---|
| Daily performance management | Paper boards, inconsistent across shifts, no escalation | Live digital SQDCP tier boards, Tier 1–3, auto-updating | |
| CAPA and deviation management | Tracked in spreadsheets, disconnected from performance | Embedded CAPA workflows, linked to KPI and owner, auto escalation | |
| Strategy deployment to shopfloor | Annual plans, lost connection to daily operations | Hoshin Kanri X-Matrix linked to team-level PDCA actions | |
| Process improvement and VSM | Static diagrams, no NVA tracking or simulation | Digital VSM with VA/NVA/PCE tracking and simulation | |
| Leadership review data | Manual compilation from ERP, LIMS, spreadsheets | Live dashboards, auto-generated, always current | |
| Root cause investigation | Ad-hoc discussions, email tracking, no structure | 5 Why, 8D, Fishbone, structured and traceable | |
| GMP compliance and audit readiness | Evidence rebuilt from emails and spreadsheets | Automatic audit trail with user, role, timestamp | |
| Live data from connected systems | Weekly exports from LIMS, SAP, MES | Direct integration with real-time data every shift |
GMP KPI Tracking Software for Pharmaceutical Manufacturing
Most operational software was never designed for GMP environments. LTS Data Point is configured for regulated pharmaceutical and biopharmaceutical operations - with audit trail, GMP KPIs, CAPA traceability, and access control built into every workflow from the start.
KPI trends, RAG status, commentary, and open actions - brought together for a complete weekly review.
Daily management, live system data, and compliant improvement tracking aligned for a digital pharma environment.
Recurring patterns across shifts and lines identified early to prevent formal deviations.
Real-time visibility of line performance, open actions, and escalations across sites from any device.
From Contract to Live Pharma Performance Management Platform in Weeks
Data Point is configured to your site - not the other way round. Pilot on one line, prove the model, scale at your pace.
Understand Your Site
We map your current KPI framework, DDS structure, CAPA process, and connected systems before configuring anything.
Configure & Customise
Your SQDCP pillars, tier boards, CAPA workflow, VSM structure, X-Matrix, and branding - built to your site's exact requirements.
Connect Your Systems
ERP, LIMS, MES, or manual inputs - your existing data sources feed dashboards automatically, no duplicate entry.
Go Live and Scale
Test, train, and go live with your dedicated LTS programme manager - then replicate across additional lines, sites, or regions at your pace.
Understand Your Site
We map your current KPI framework, DDS structure, CAPA process, and connected systems before configuring anything.
Configure & Customise
Your SQDCP pillars, tier boards, CAPA workflow, VSM structure, X-Matrix, and branding - built to your site's exact requirements.
Connect Your Systems
ERP, LIMS, MES, or manual inputs - your existing data sources feed dashboards automatically, no duplicate entry.
Go Live and Scale
Test, train, and go live with your dedicated LTS programme manager - then replicate across additional lines, sites, or regions at your pace.
Everything You Need to Deploy and Scale Pharmaceutical Performance management software
Data Point is designed for real-world pharmaceutical deployment - with guided onboarding, GMP-ready configuration, secure cloud infrastructure, and dedicated LTS support built in from day one.
Implementation & Enablement
Everything you need to deploy Data Point in a regulated pharmaceutical manufacturing environment.
- Guided onboarding with dedicated LTS programme management
- Bespoke SQDCP / SQODCPI framework and tier board configuration
- System integration setup: ERP, LIMS, MES, and manual data inputs
- Role-based training for shift leaders, QA, and site management
- Adoption support across lines, departments, and sites
Enterprise Delivery & Scalability
Secure, scalable, and built for enterprise pharmaceutical operations across multiple sites.
- Secure cloud hosting, automated backups, and platform maintenance
- Unlimited users across teams, divisions, and locations
- Mobile-ready access for distributed leadership teams
- Continuous software updates and new capability releases
- Dedicated LTS programme manager supporting your account long-term
Who Uses Data Point
Built for Every Role in Pharmaceutical Operations
Data Point serves every leader in the pharma performance chain - from the site director setting OEE targets to the QA manager closing CAPA actions.
Customer Success Story
“By providing a central location to input, analyse and share our KPIs, 'Data Point' enables site management to more easily focus on the entire business as a team. Its ability to allow automated data entry and trend analysis gives us more time for improvement rather than just reporting numbers. Combined with a disciplined approach within our SQDC meeting process, I believe 'Data Point' will help us continually focus on key issues and drive business excellence in all areas.”
Marc Robinson
Director, Global Operational Excellence


PCI Pharma Streamlined Continuous Improvement with SQCDP Boards
PCI Pharma Services Bridgend site faced significant challenges in managing their daily stand-up meetings and reviewing crucial metrics impacting their production and operations plans. Relying heavily on paper-based systems and spreadsheets during Lean Daily Management Meetings, they struggled to effectively track and analyse their Safety, Quality, Delivery, Cost, and People (SQDCP) key performance indicators (KPIs). To overcome these hurdles and propel their continuous improvement journey, PCI Bridgend ...
LTS Data Point in Pharmaceutical Operations - Real Outcomes
See how pharmaceutical manufacturers and biopharmaceutical organisations have used LTS Data Point to improve performance, compliance, and operational visibility.
Run your pharma site performance reviews on a platform built for it
Fill out the form below to see how Data Point connects OEE, CAPA, batch yield, and GMP compliance in one live platform — built for regulated pharmaceutical manufacturing sites.












